A major step in preclinical drug development is demonstrating the antitumor activity of a novel agent in animal models, alongside with the choice of the optimal dosage and treatment schedule that balance efficacy and safety. Our in vivo services cover every critical checkpoint of this process, from model selection, toxicity profiling and efficacy evaluation to deep tumour characterisation.
Our in vivo service offerings include:
01 – Model selection and study design
- Expert guidance on indication & model choice
Selecting the right preclinical tumor model is essential for generating relevant and predictive data. Based on our expertise, we provide guidance to help you choose the most appropriate models based on your compound’s mechanism of action and the cancer types you wish to target. Whether you are developing immunotherapies, small molecules, or biologic agents, we can help you identify the best indication and tumor model to put all the chances on your side for the success of your preclinical studies. Our selection includes both human-derived xenograft (CDX) models, syngeneic murine models, and patient-derived-xenografts (PDX) models.
- A catalogue of 150+ characterised tumour models
Our database includes more than 100 human-derived CDX models and 40 syngeneic mouse models. These models include a variety of solid tumors, including breast, lung, colorectal, and melanoma, as well as hematologic malignancies (e.g., leukemia and lymphoma). These well-established models offer a robust platform for researching the toxicity, immunological responses, and effectiveness of drugs in various tumor types.
- Secondary resistance models to reference therapies
As resistance to treatment is one of the most significant challenges in oncology, we offer secondary resistance models that mimic the acquired resistance seen in patients who initially respond to treatment but eventually relapse. Our unique CDX and syngeneic models of secondary resistance to reference therapies (e.g., immune checkpoint inhibitors, chemotherapy) allow to investigate the mechanisms of resistance and enable the evaluation of new therapies that may overcome this challenge.
- On-demand model development
We also offer custom development of new models, and in particular resistant tumor models to a standard of care of interest for you or to your own therapy to evaluate specific mechanisms of resistance in immunotherapy and chemotherapy. These models are ideal for testing novel compounds that aim to reverse resistance or sensitize tumors to current therapies.
02 – Toxicity assessment
- Systemic and clinical toxicity in rodents
A crucial aspect of preclinical development is evaluating the toxicity profile of preclinical compounds. Our rodent studies allow assessing the systemic toxicity of your compounds, including both acute and chronic exposure scenarios, to identify any adverse effects or organ toxicity that may arise.
- Haematological toxicity
We also evaluate hematological toxicity, analyzing potential impacts on blood cell counts and immune function, helping you assess the overall safety of your compound before moving forward to clinical trials.
- Metabolic toxicity
Assessment of key metabolic parameters via Vetscan VS2 SCIL® (including ALT, albumin, GGT, alkaline phosphatase, urea, cholesterol, biliary acids and total bilirubin) to evaluate potential organ toxicity.
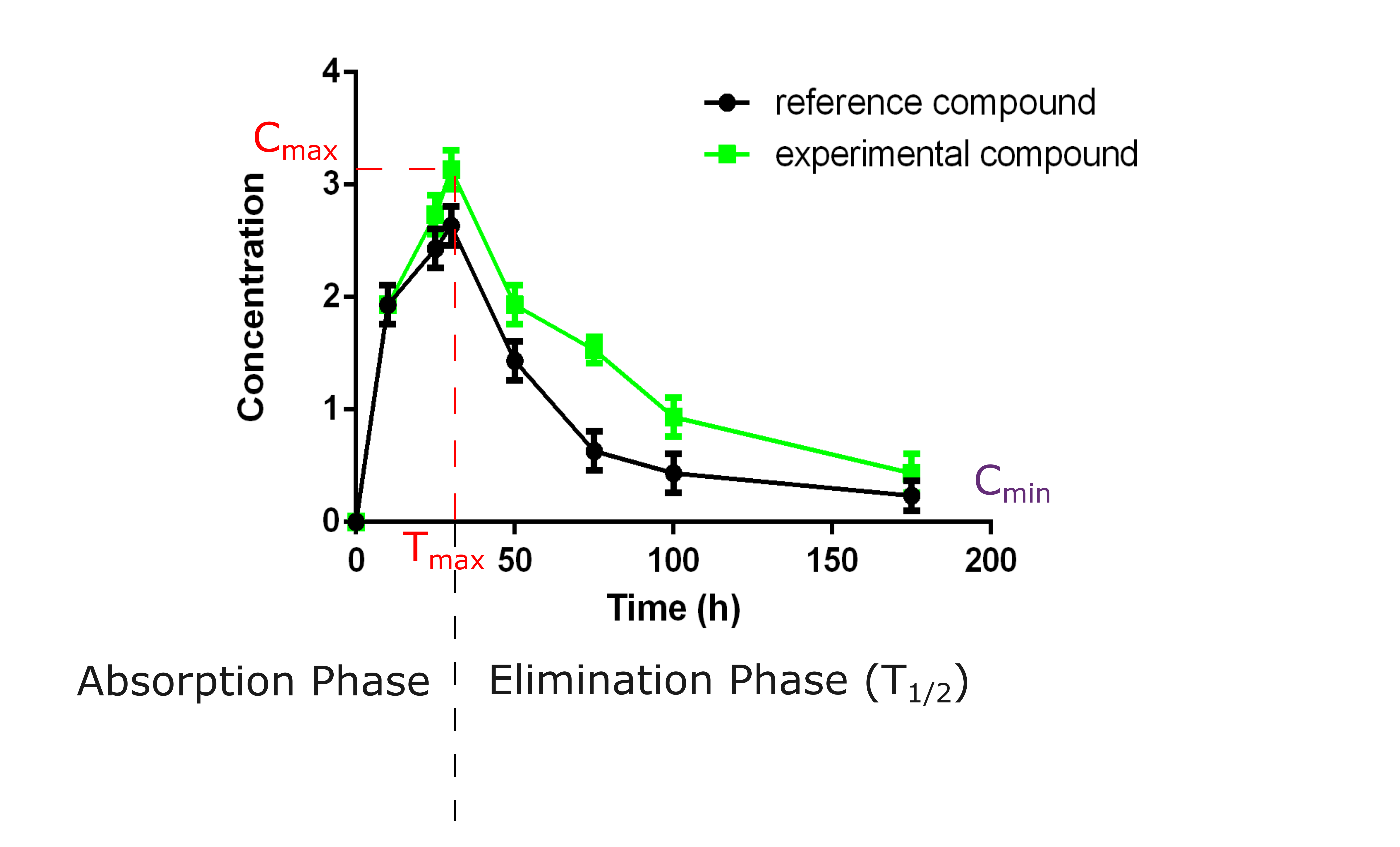
03 – Pharmacokinetics (PK)
- PK studies in mouse and rat
The understanding of the PK properties of your compound is essential for designing effective dosing regimens in clinical settings. We conduct PK studies in mice and rats to assess how your compound is absorbed, metabolized, and eliminated from a whole organism.
- Half-life determination & dosing optimisation
After an analysis by mass spectrometry, the results will help define the half-life of your compound, a critical point for optimizing the dosing schedule and ensuring therapeutic efficacy while minimizing toxicity.
04 – Antitumour efficacy
- Efficacy studies in human & murine models
Tumour models (representative of solid tumours and haematological malignancies) are implanted subcutaneously or orthotopically by injection of cell suspensions, tumour fragments or dissociated tumour cells (with or without Matrigel). Treatments are administered intraperitoneally, intravenously, orally, by direct tumour injection or continuous infusion, up to 7 days a week.
Results include: tumour volume evolution over time, dissemination to other localisations (assessed by imaging or macroscopic/microscopic analyses), and survival analyses based on predefined endpoints.
- Combination studies & comparison with standards of care
To evaluate the relative efficacy of your novel compound, we can conduct combination studies or comparative studies using gold standard treatments in various cancer types. Whether comparing a novel agent with a chemotherapeutic, immune checkpoint inhibitor, or targeted therapy, these studies allow us to assess how your compound performs in combination or as an alternative to current therapeutic options, providing valuable data on synergistic effects, treatment resistance, and potential patient benefit.
05 – Tumour and sample analysis
- Tumour and organ sampling
Tumour and organ samples can be fixed in formaldehyde (provided as such, as paraffin blocks or sections on slides), frozen at -80°C, or snap-frozen in liquid nitrogen for metabolomic studies. Blood and tissue sampling can be performed at multiple time points for complementary analyses by Antineo or your preferred service provider.
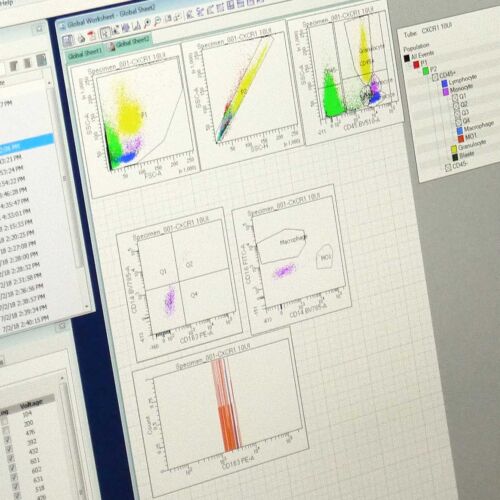
- Immunophenotyping of the tumour microenvironment
Understanding the tumor microenvironment (TME) is important for developing therapies that not only target tumor cells but are also able to modulate the immune system within the tumor. We offer immunophenotyping studies of the TME, including the analysis of immune cell populations, cytokine profiles, and immune checkpoint markers in both tumor tissue and peripheral blood. These studies can help you understand how your therapy affects the immune landscape and its potential to enhance antitumor immunity.
- Flow cytometry analysis
Tumour samples can be analysed by flow cytometry to determine the expression level of markers of interest or for in-depth immunophenotyping of the TME using our high-dimensional platform with up to 29-marker panels.
Would you like to discuss your in vivo study design or find out which models and services best fit your compound? Our scientific team is here to help, contact us.

 Antineo
Antineo Preclinical services
Preclinical services Tumour models
Tumour models Our Strengths
Our Strengths News & Events
News & Events